Page 73 - 《中国药科大学学报》2026年第2期
P. 73
第 57 卷第 2 期 吴 杰,等:Asundexian 衍生物的设计、合成及活性评价 199
CF 3 MeO
O C N N N CF 3 OMe
NH 2 HN H N CF 3 N N HO B OMe N
Br a Br b Br N N 24 Br OH 26 N N
c d
OMe
Cl Cl Cl Cl
Cl
21 22 23 25 27
CF 3 O CF 3 CF 3
N O Br N O N O
N O N O N OH
e N NH 29 N N g N N
f O O
OMe OMe OMe
Cl Cl Cl
28 30 31
CF 3 CF 3
N O H N O H
N N F N N F
N N O N N O H
O O N
N R 1 S R 3
O H O O
Cl FA-1-FA-6 Cl
12a-12b FB-1-FB-2
4a-4f
CF 3
N O
N OH
N N
CF 3 O
N O 16a-16b O 9a-9b N CF 3 O
N H F H
N
N N O Cl N N N N F
O 31 O
N O S R 2
O N
R 4 O O
Cl 20a-20b Cl
FC-1-FC-2 FB-3-FB-4
CF 3
N O H
N N F
N N H
O N
N R 5
O O H
Cl
FD-1-FD-2
S O
R 1a = N R 1d = R 2a = R 3a = R 4a = O S O R 5a = O
N N
O O
O S
R 1b = N R 1e = R 2b = R 3b = N R 4b = N R 5a = O S O
N N O S O
O
R 1c = N R 1f =
N
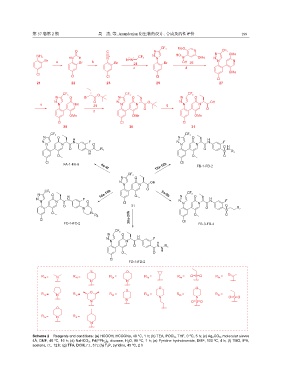
Scheme 2 Reagents and conditions: (a) HCOOH, HCOONa, 40 ºC, 1 h; (b) TEA, POCl 3 , THF, 0 ºC, 5 h; (c) Ag 2 CO 3 , molecular sieves
4Å, DMF, 40 ºC, 16 h; (d) NaHCO 3 , Pd(PPh 3 ) 4 , dioxane, H 2 O, 95 ºC, 1 h; (e) Pyridine hydrobromide, DMF, 100 ºC, 4 h; (f) TMG, IPA,
acetone, r.t., 12 h; (g) TFA, DCM, r.t., 3 h; (h) T 3 P, pyridine, 45 ºC, 2 h